 
The breakthrough that produced the earliest form of the modern Li-ion battery was made by British chemist M. Research on rechargeable Li-ion batteries dates to the 1960s one of the earliest examples is a CuFĢ/Li battery developed by NASA in 1965. Main article: History of the lithium-ion battery Strategies include aqueous lithium-ion batteries, ceramic solid electrolytes, polymer electrolytes, ionic liquids, and heavily fluorinated systems. Research has been under way in the area of non-flammable electrolytes as a pathway to increased safety based on the flammability and volatility of the organic solvents used in the typical electrolyte. Research areas for lithium-ion batteries include extending lifetime, increasing energy density, improving safety, reducing cost, and increasing charging speed, among others. Both environmental issues have encouraged some researchers to improve mineral efficiency and alternatives such as iron-air batteries. Moreover, both lithium and other key strategic minerals used in batteries have significant issues at extraction, with lithium being water intensive in often arid regions and other minerals often being conflict minerals such as cobalt. Improperly recycled batteries can create toxic waste, especially from toxic metals and are at risk of fire. 
Lithium ion all solid state batteries are being developed to eliminate the flammable electrolyte. Much development has made progress in manufacturing safe lithium-ion batteries. Lithium-ion batteries can be a safety hazard if not properly engineered and manufactured since cells have flammable electrolytes and if damaged or incorrectly charged, can lead to explosions and fires. The first prototype of the modern Li-ion battery, which uses a carbonaceous anode rather than lithium metal, was developed by Akira Yoshino in 1985, which was commercialized by a Sony and Asahi Kasei team led by Yoshio Nishi in 1991. John Goodenough expanded on this work in 1980 by using lithium cobalt oxide as a cathode. Stanley Whittingham discovered the concept of intercalation electrodes in the 1970s and created the first rechargeable lithium-ion battery, which was based on a titanium disulfide cathode and a lithium-aluminum anode, although it suffered from safety issues and was never commercialized. NMC and its derivatives are widely used in the electrification of transport, one of the main technologies (combined with renewable energy) for reducing greenhouse gas emissions from vehicles. Lithium iron phosphate ( LiFePOģ-based lithium rich layered materials, LMR-NMC), and lithium nickel manganese cobalt oxide ( LiNiMnCoOĢ or NMC) may offer longer lives and may have better rate capability. Handheld electronics mostly use lithium polymer batteries (with a polymer gel as electrolyte), a lithium cobalt oxide ( LiCoOĢ) cathode material, and a graphite anode, which together offer a high energy density. Cells can be manufactured to prioritize either energy or power density. The anode or negative electrode is usually graphite, although silicon-carbon is also being increasingly used. Most commercial Li-ion cells use intercalation compounds as the active materials. Ĭhemistry, performance, cost and safety characteristics vary across types of lithium-ion batteries. Compared to other rechargeable battery technologies, Li-ion batteries have high energy densities, low self-discharge, and no memory effect (although a small memory effect reported in LFP cells has been traced to poorly made cells). It also sees significant use for grid-scale energy storage and military and aerospace applications. It is the predominant battery type used in portable consumer electronics and electric vehicles.  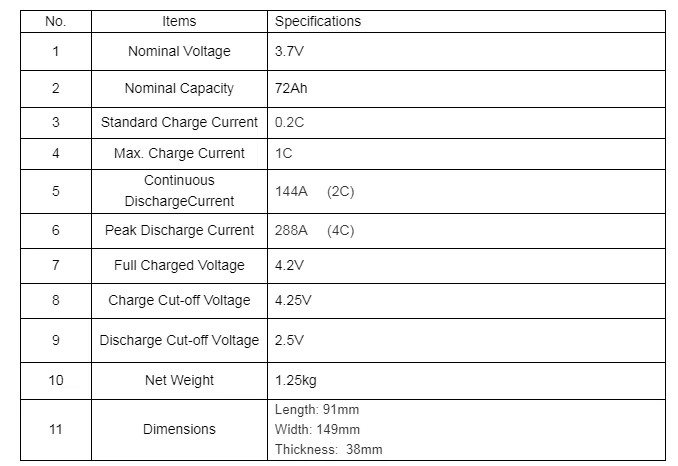
The electrolyte is typically a lithium salt in an organic solvent. The cathode (positive electrode) is typically a metal oxide. The anode (negative electrode) of a conventional lithium-ion cell is typically graphite made from carbon. A 3.6v Li-ion battery from a Nokia 3310 mobile phoneġ00–265 Wh/kg (0.360–0.954 MJ/kg) Ġ.35% to 2.5% per month depending on state of charge ģ.6 / 3.7 / 3.8 / 3.85 V, LiFePO4 3.2 V, Li4Ti5O12 2.3 VĪ lithium-ion or Li-ion battery is a type of rechargeable battery which uses the reversible reduction of lithium ions to store energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
